Accelerating Healthcare Approvals: 3 AI Strategies That Turn Weeks Into Hours
Ask any marketing leader in pharma, medtech, or biotech what slows down campaigns. The answer comes immediately: the approval process.
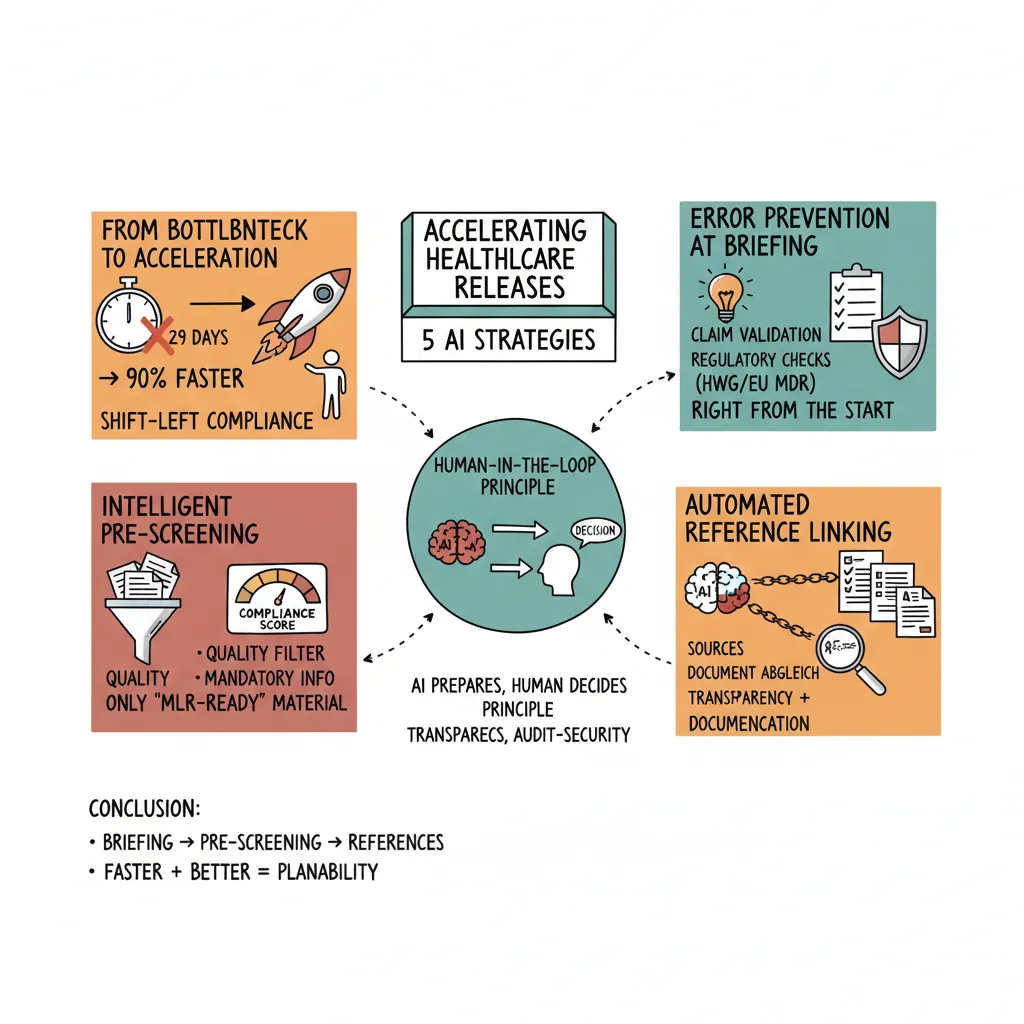
29 business days for a single marketing asset. In some companies, even 12 weeks. Campaigns miss market windows, competitors gain ground, and your marketing team burns energy in the endless ping-pong between Medical, Legal, and Regulatory.
The problem isn't with the review departments. It's in the process itself.
Why the MLR Review Becomes a Bottleneck
The traditional approval process follows a pattern unchanged for decades: Marketing develops a campaign idea, creates briefings, commissions copy and visuals. Only when everything is finished does the material go into MLR review (Medical, Legal, Regulatory).
That's when problems surface:
- Claims without source references from prescribing information or clinical studies
- Statements that exceed approved indications or intended purposes
- Missing mandatory disclosures required by FDA, EMA, or local regulations
- Unbalanced benefit-risk presentation
- Unclear distinction between HCP and consumer-facing materials
The result: queries, revision cycles, re-reviews. An asset often goes through multiple correction cycles before approval. Each cycle costs a week or more.
Turnaround time is the symptom. The root cause: Compliance comes too late.
| Industry | Typical Challenges |
|---|---|
| Pharma | FDA/EMA mandatory disclosures, PI alignment, RX vs. OTC rules |
| MedTech | EU MDR Article 7, CE marking in advertising, intended use claims |
| Biotech | Complex scientific claims, off-label risks |
3 AI Strategies That Transform the Approval Process
Artificial intelligence cannot replace the MLR review, but it can radically shorten it. Not by automating the final review, but by shifting compliance logic earlier in the process.
Three approaches have proven particularly effective:
1. Compliance Checking at Briefing: Prevent Errors Before They Happen
The biggest leverage point is at the beginning. If a briefing already contains compliance risks, the problem propagates through the entire process: from agency to creative to final rejection by Medical.
What AI-powered compliance checking delivers here:
- Real-time claim validation: As soon as you formulate a key message, AI automatically checks against the claims library. "Fast-acting relief" sounds good, but is it supported by prescribing information or study data? AI provides immediate feedback before the briefing goes to the agency.
- Regulatory conformity check: AI identifies during the planning phase whether planned statements require mandatory disclosures, whether under FDA regulations for pharmaceuticals or EU MDR Article 7 for medical devices. An OTC product claim is subject to different rules than RX advertising to HCPs or advertising for a Class IIa medical device.
- Audience routing: Depending on whether material targets healthcare professionals or consumers, different requirements apply. AI flags the relevant regulations early, before creative work begins.
The effect: Instead of "finished but wrong," you get "right from the start." Your team receives immediate feedback on ideas and can design campaigns that are compliance-ready, without unpleasant surprises weeks later in MLR review.
Practical example: A briefing for a medical device includes the planned claim "Clinically superior outcomes." AI immediately flags: Comparative advertising requires direct comparative studies under EU MDR. Alternative wording is suggested. Result: No rejection from Regulatory review.
2. Pre-Screening Before MLR Review: The Intelligent Quality Filter
Even when the briefing was clean: Errors creep in during execution. Copy gets adjusted, visuals swapped, wording changed. The finished material no longer matches the original plan.
This is where pre-screening comes in: An AI-powered preliminary check before material even reaches Medical, Legal, and Regulatory.
What AI-powered pre-screening delivers:
- Automatic claim detection: AI identifies all promotional statements in the material and flags them, including categorization by claim type (efficacy, safety, comparative). No claim slips through unnoticed.
- Compliance score: Each asset receives a rating showing how "MLR-ready" it is. A 95% score signals: minor adjustments needed. A 60% score means: back to the agency before a reviewer invests time.
- Mandatory disclosure check: Missing required statements? Is the CE marking compliant? Is the warning correctly placed? Is the benefit-risk information balanced? AI flags these issues automatically.
- Similarity check with DAM: Is the material similar enough to already approved assets to justify an expedited review? AI compares against the database of approved materials and identifies reusable elements.
The effect: MLR teams receive only material that meets basic requirements. Instead of wasting time on obvious errors, Medical Reviewers can focus on substantive nuances and scientific accuracy.
| Without Pre-Screening | With AI Pre-Screening |
|---|---|
| Multiple review cycles | 1-2 review cycles |
| First errors discovered at MLR | Errors fixed before submission |
| Reviewers frustrated | Reviewers focused |
| Turnaround: 4-12 weeks | Turnaround: drastically reduced |
3. Automatic Reference Linking: End the Source Hunt
An underestimated time drain in the approval process: manual source attribution. Every promotional claim needs evidence from prescribing information, clinical studies, technical documentation, or approved reference documents.
In practice, this often looks like: The Medical Reviewer reads a statement, opens the prescribing information or study database, searches for the relevant passage, verifies alignment, documents the reference. Multiplied by dozens of claims per asset and hundreds of assets per year, this adds up to weeks of lost productivity.
What AI-powered reference linking delivers:
- Automatic source linking: AI automatically assigns the appropriate reference to each promotional statement, with a direct link to the original source. "47% symptom reduction" is automatically linked to the corresponding study passage.
- Document alignment: Every claim is checked against current prescribing information, instructions for use (IFU), or technical documentation. Does the wording deviate? Is the statement covered by the approved indication or intended use? AI flags discrepancies immediately.
- Claims library with versioning: A central database maintains all approved statements with their sources. When prescribing information updates or regulatory changes occur, affected claims are automatically flagged. Critical for compliance across the entire content lifecycle.
- Audit trail for regulatory requirements: For each reference assignment, AI documents which source was selected and why. This creates transparency for audits, essential for FDA-regulated markets (21 CFR Part 11) and EU compliance (Annex 11).
The effect: Medical Reviewers don't need to search; they just need to confirm. AI delivers the source work on a silver platter. This not only saves time but also reduces errors from overlooked or outdated references.
The Combination Makes the Difference
Viewed individually, each of these approaches saves time. Together, they fundamentally change the logic of the approval process:
| Traditional Process | AI-Powered Process |
|---|---|
| Compliance check at the end | Compliance check from the start |
| Serial review loops | Parallel pre-checks |
| Manual source search | Automated reference linking |
| Same capacity, more assets = longer queues | Scalable without proportional delay |
The Bottom Line
The MLR review will remain a critical control point. Medical, Legal, and Regulatory teams ensure that healthcare marketing meets regulatory requirements and doesn't put patients at risk. This responsibility cannot be delegated to AI.
But what can be delegated: the preparatory work, the obvious error checking, the tedious source attribution. AI handles these tasks faster, more consistently, and without fatigue.
The result: Marketing teams gain predictability. MLR teams gain focus. And campaigns reach the market when they're actually needed.
Three key takeaways:
- ✓ Compliance checking at briefing prevents errors from happening in the first place
- ✓ Pre-screening before MLR ensures only ready material gets reviewed
- ✓ Automatic reference linking eliminates the biggest manual time drain
Marketing teams in pharma, medtech, and biotech that implement these approaches don't just gain time. They gain predictability, reduce frustration, and can finally launch campaigns when the market needs them.
Sources
- LTIMindtree (2025). Speed to Market: How Gen AI accelerates MLR Reviews for Pharma. https://www.ltimindtree.com/blogs/speed-to-market-how-gen-ai-accelerates-mlr-reviews-for-pharma/
- Klick Health (2025). Survey: 65 Percent of Pharma Marketers Distrust AI for Creating Regulatory Compliance Submissions. https://www.klick.com/
- Cognizant (2017). Taking Control of the MLR Review Process. Cognizant Whitepaper.
- PwC & Aquent (2025). The Impact of Content Automation on Project Timelines in Life Sciences.
