The Complete Guide to MLR Review

MLR review is the single biggest bottleneck standing between your pharma marketing team and faster campaign launches. Short for Medical, Legal, and Regulatory review, it is the process life sciences companies use to ensure every piece of promotional content is compliant before it reaches the market. This guide covers what MLR review means, how the process works, why it consistently breaks down, and how AI is changing what is possible for life sciences content teams.
What Is MLR Review?
What Does MLR Stand For?
MLR stands for Medical, Legal, and Regulatory: the three functions responsible for evaluating promotional content in pharmaceutical, biotech, and medical device companies. The MLR full form reflects its cross-functional nature: no single team owns the review; all three must sign off.
In medical and pharmaceutical contexts, the MLR full form is Medical, Legal, and Regulatory. You will also encounter the term written as MMLR (Marketing, Medical, Legal, Regulatory) in some organisations. The meaning is the same regardless of the acronym used.
Medical, Legal, and Regulatory (MLR) review is the formal process these companies use to evaluate promotional and marketing materials before they reach healthcare professionals or patients. Every claim, every visual, every disclaimer in a piece of promotional content must pass through this multi-disciplinary review to confirm it is scientifically accurate, legally sound, and fully compliant with applicable regulations.
The three disciplines each serve a distinct function. The medical reviewer checks that clinical claims are supported by evidence and consistent with approved labeling. The legal reviewer assesses liability exposure, intellectual property considerations, and comparative claims. The regulatory reviewer verifies alignment with the relevant regulatory frameworks: in the US, primarily FDA rules under 21 CFR Part 202 (Prescription Drug Advertising) and FDA guidance on fair balance and substantiation requirements; in Europe, Directive 2001/83/EC for medicinal products, EU MDR provisions for medical device claims, and national codes such as Germany's Heilmittelwerbegesetz (HWG).
MLR review applies to a broad range of content: sales aids, digital advertising, social media posts, medical congress materials, patient-facing websites, and email campaigns. If it promotes a prescription drug, biologic, or regulated medical device, it goes through MLR regardless of channel or format.
The process is also referred to as Promotional Review Committee (PRC) review. The terminology varies, but the purpose is the same: ensure no promotional claim reaches the public without proper scrutiny.
Why MLR Review Breaks Down
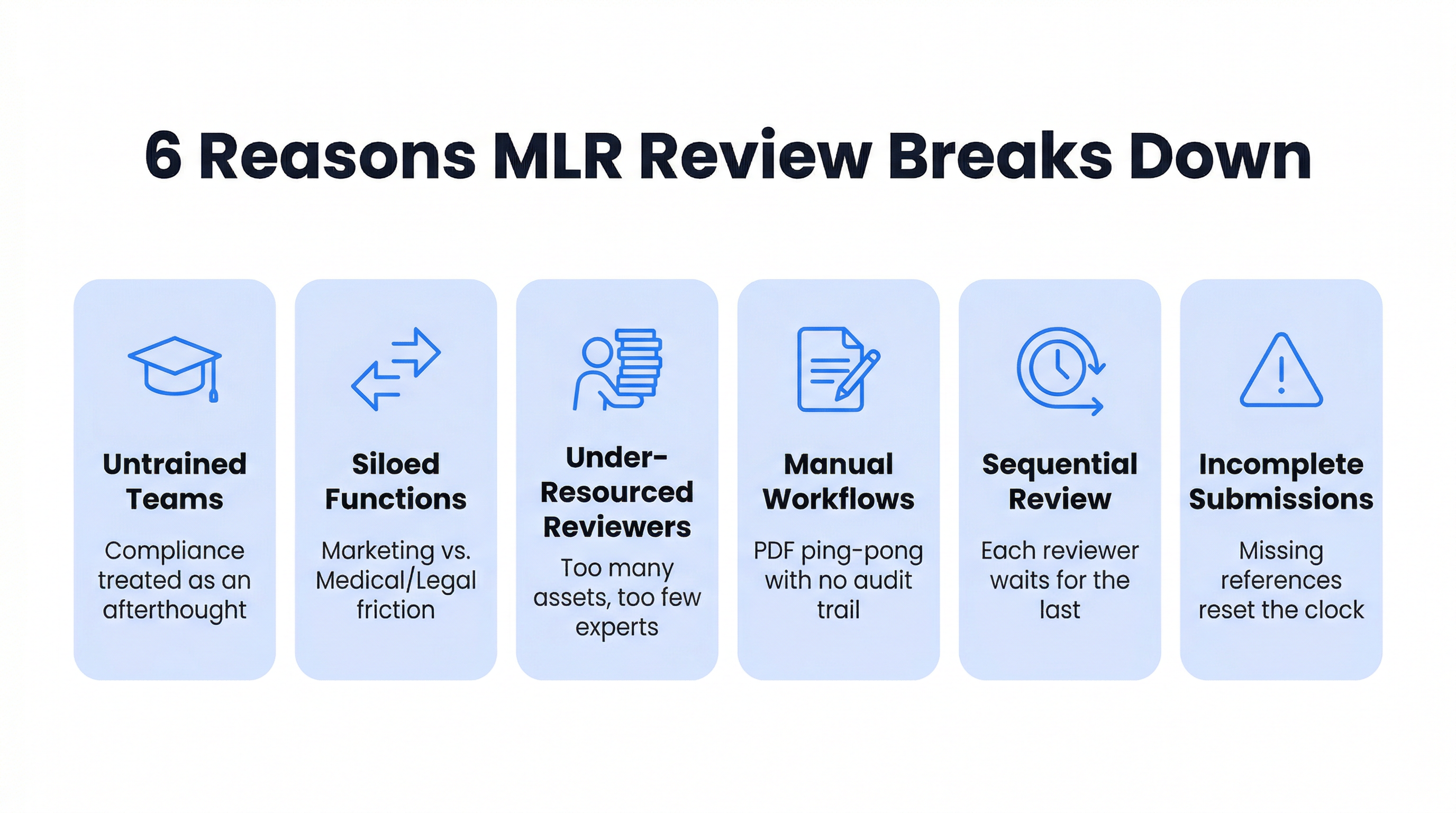
If your campaigns consistently launch late, the MLR process is likely where time is being lost. This is one of the most widely shared frustrations across pharma and medtech marketing teams. Understanding exactly where the friction comes from is the first step to fixing it.
After working closely with life sciences marketing and regulatory teams across the DACH region, we have seen the same patterns repeat regardless of company size. The dysfunction is rarely unique. It is structural.
Marketing teams are not trained for compliance
Junior marketers in particular operate with pure marketing logic and very little regulatory awareness. Compliance is treated as a blocker rather than a built-in quality standard. Content gets created without any consideration for MLR requirements from the start, which means the first time a compliance lens is applied to an asset is during the review itself. That is too late.
I've sat in on briefings where the campaign concept was basically finished before anyone had thought to ask whether the central claim was even approvable. The whole thing had to be restructured from scratch. That was a four-week delay on a campaign that took two days to concept.
— Max, Co-Founder, Caidera
Marketing and Medical/Legal work against each other
Cross-functional workflows are barely coordinated in most organisations. Marketing operates at speed with commercial deadlines. Medical and Legal operate with caution, serving risk and accountability. Neither team fully understands the other's constraints, and there is rarely a shared process that bridges them. The result is a handoff that feels adversarial on both sides.
The marketing team thinks Legal is being obstructive. Legal thinks Marketing is being reckless. Both are right about each other. Nobody has designed a process that serves both goals at once, so they just fight it out asset by asset.
— Max, Co-Founder, Caidera
Medical and Legal teams are chronically under-resourced
These departments carry the review burden with a fraction of the headcount needed to match content volume. A single regulatory reviewer may be responsible for dozens of assets in flight simultaneously. Bottlenecks accumulate fast, cascading into significant marketing delays. Those delays force rescheduling, which creates compounding extra work for everyone downstream.
The review is still largely manual and document-based
Content still circulates via PDFs with dozens of inline comments in most organisations we speak to. There is no structured, auditable workflow. Just document ping-pong. This creates high effort per review cycle for already stretched expert teams, with no visibility into status, no version control, and no way to learn from previous feedback.
We spoke with one team that was routing assets through six email threads simultaneously. Medical had one version. Legal had another. Nobody was sure which was current. The regulatory reviewer was working from a PDF that was two drafts behind.
— Max, Co-Founder, Caidera
Sequential review instead of parallel
When medical, legal, and regulatory reviewers work through the same asset one after another rather than simultaneously, you add the full duration of each reviewer's cycle time to the total. A process that might take one week in a parallel structure can easily extend to three or more when each reviewer works sequentially. That cost compounds across a full content calendar.
Incomplete submissions that reset the clock
When an asset arrives without a complete reference pack (missing citations, unlinked claims, or absent mandatory statements), the reviewer either rejects the submission outright or flags it for resubmission. Either way, the clock resets. Most teams underestimate how often this happens and how much cumulative time it costs.
At Caidera, the most common issue we observe is not that reviewers are slow. It is that the same compliance errors appear repeatedly in submissions, forcing multiple rounds of feedback on problems that could have been caught before the asset ever entered the queue. Teams that introduce pre-submission compliance checks typically cut their revision rounds significantly within the first two months.
The MLR Review Process: A Step-by-Step Guide
Understanding how the MLR review process works in practice helps your team design a faster, cleaner workflow.
The six stages of a typical MLR cycle
A typical MLR cycle moves through six stages. First, the marketing or content team creates the asset and packages it with a reference pack supporting every claim made. Second, the asset is submitted to the review queue, usually via a promotional content review system or workflow platform. Third, medical, legal, and regulatory reviewers each assess the asset, either sequentially or in parallel depending on the organisation's setup. Fourth, reviewers return consolidated feedback. Fifth, the content team revises and resubmits. Sixth, once all three functions sign off, the asset is cleared for publication.
What reviewers actually check
What gets reviewed goes beyond the written copy. Reviewers check factual claims against approved labeling and published clinical data, assess fair balance between benefit and risk statements, verify that disclaimers and mandatory statements are present and correctly formatted, and evaluate visual elements including images, graphs, and infographics for accuracy and compliance.
How long does an MLR review take?
The timeline varies significantly by organisation and asset complexity. At mid- and large-sized MLR review pharma companies, review cycles commonly stretch to 50 to 60 days per content piece.1 In the European market, content approval takes 20 days on average, with a mean of 1.3 review cycles per asset.2
In practice, the most significant delays rarely come from the review itself. Incomplete reference packs, fragmented feedback, unclear versioning, and sequential review structures all add days or weeks to each cycle. Most of these variables sit within your team's control.
The Key Stakeholders in MLR Review
Every MLR review involves three core functions, each evaluating the same piece of content through a different lens.
The medical reviewer
The medical reviewer is typically a physician, pharmacist, or medical science liaison. Their focus is scientific accuracy: are the clinical claims consistent with the approved label, supported by peer-reviewed data, and free from off-label implications? They also assess fair balance: whether the material presents risk information with comparable prominence and readability to benefit claims, consistent with the FDA's fair balance standard.
The legal reviewer
The legal reviewer looks at the asset from a liability and intellectual property perspective. They check comparative claims, assess whether any language could be construed as a warranty or guarantee, review endorsement language, and ensure the material does not create grounds for a false advertising claim. In regulated markets, they also verify that disclaimers meet jurisdictional requirements.
The regulatory reviewer
The regulatory reviewer is the final checkpoint before publication. Their job is to ensure the asset aligns with the relevant regulatory framework: FDA rules under 21 CFR Part 202 in the US, Directive 2001/83/EC for medicinal products in Europe, and national codes such as Germany's HWG. They check that mandatory statements are present and that the overall presentation does not create a misleading impression of the product.
The Promotional Review Committee (PRC)
These three reviewers often operate as a Promotional Review Committee (PRC): a standing cross-functional team that meets on a regular cadence to process submissions. Some organisations run PRC meetings weekly; others use asynchronous digital workflows. The structure varies, but the accountability is the same.
Marketing teams are participants in this process, not bystanders. In our experience working with life sciences teams, the teams that move fastest through MLR are not the ones with the most lenient reviewers. They are the ones that arrive at review with fewer errors to catch.
Ready to reduce your MLR revision rounds? See how Caidera flags compliance issues before content reaches your review queue. Book a demo →
How to Optimise Your MLR Review Process
Improving MLR process efficiency does not require replacing your review team or overhauling your entire content stack. Most organisations can meaningfully reduce cycle times by fixing the inputs, the structure, and the feedback loop.
Standardise your submission requirements
The most impactful single change most teams can make is enforcing a complete, consistent submission package before any asset enters the review queue. This means a finalised asset with all claims clearly linked to supporting references, a cover sheet identifying the asset type, target audience, and intended channel, and a checklist confirming that mandatory statements and fair balance requirements have been addressed. Reviewers who receive complete submissions spend their time reviewing, not chasing missing materials.
Move from sequential to parallel review
There is no regulatory requirement that medical, legal, and regulatory reviewers must work in sequence. If your organisation runs sequential reviews by default, challenge that assumption. Parallel review, where all three functions assess the same version simultaneously, can cut total review time significantly. In most cases it does not meaningfully increase the burden on individual reviewers. The precondition is a shared platform that allows multiple reviewers to annotate the same asset without creating version conflicts.
Set SLAs for every review stage
Undefined timelines invite drift. Set explicit turnaround expectations for each reviewer function: five business days for standard assets, two for expedited, for example. Make those SLAs visible to all stakeholders. In most organisations, when reviewers know a deadline is being tracked and visible, response times tend to improve.
Build a centralised claims library
A claims library is a repository of pre-approved language: validated efficacy statements, approved risk disclosures, cleared comparative claims, and mandatory copy elements. When your content team drafts against approved claims from the outset, the number of novel compliance issues entering review drops substantially. Regulatory and medical reviewers have already signed off on the building blocks; they only need to assess how those blocks have been assembled.
Bring reviewers in earlier
The most expensive revision is one that requires restructuring a nearly-finished asset. Many organisations involve MLR only at the end of the content creation process, after creative and messaging work is complete. Introducing a lightweight regulatory checkpoint at the briefing stage, before significant production effort has been invested, surfaces structural compliance issues early when they are cheap to fix.
The best-run MLR teams we have seen brief their regulatory reviewer at the concept stage. Not to approve the campaign, just to flag whether the core claim is approvable at all. It takes 30 minutes and saves weeks.
— Max, Co-Founder, Caidera
Measure what matters
Effective MLR review management starts with measurement. Monitoring average cycle time per asset type, revision round frequency, and the most common rejection reasons gives your team the data to identify where time is actually being lost and where intervention will have the most impact.
For more on this topic, read our guide on why email-based approvals sabotage marketing workflows and AI strategies for accelerating healthcare approvals.

What to Look For
As the pharmaceutical promotional review process moves off email and into dedicated platforms, the choice of tooling has a direct impact on cycle times, audit readiness, and reviewer experience. Not all MLR review software is built the same way. For life sciences teams, the differences matter.
Workflow automation
This is the baseline capability any serious MLR platform should offer: configurable routing rules that send assets to the right reviewers automatically, deadline tracking with notifications, and status visibility for all stakeholders in real time. If your platform requires a coordinator to manually move assets between reviewers, it is adding process overhead rather than removing it.
Annotation and commenting tools
Reviewers need to be able to mark up assets in context: highlighting specific text, attaching comments to individual claims, and threading replies, rather than describing changes in a separate email. When feedback is anchored to the asset itself, revision briefs are clearer and revision rounds are shorter.
A full audit trail
Under 21 CFR Part 11 in the US and equivalent frameworks in regulated markets, electronic records used in promotional review must include a complete, attributable, and legible audit trail. Any platform that cannot generate an immutable record of the review and approval history should not be handling regulated promotional content.
Analytics and reporting
The ability to measure average cycle time by asset type, identify which reviewers or stages are creating bottlenecks, and track revision frequency over time gives operations and medical affairs leaders the data to drive continuous improvement. Without this visibility, optimisation efforts are based on anecdote rather than evidence.
Ease of use for non-technical reviewers
This is frequently underweighted during platform selection and frequently surfaces as a friction point in post-implementation feedback. Medical, legal, and regulatory reviewers are subject-matter experts, not software users by trade. A platform that requires significant training or creates friction in the review experience will face adoption problems. Adoption problems translate directly into workarounds, and workarounds translate directly into compliance gaps.
Integration with existing tools
The ability to ingest assets from the systems where they are created, whether that is a DAM, a CMS, or a creative production platform, without requiring manual uploads and reformatting eliminates a step that adds time without adding value.
When evaluating MLR review software, the core question is not which platform has the most features. It is which platform will actually be used, consistently, by your medical, legal, and regulatory reviewers, and which will give your compliance team the audit-ready documentation they need when it matters.
The Role of AI in MLR Review
Artificial intelligence is changing how life sciences companies approach the front end of the MLR process. Not by replacing human reviewers, but by improving the quality of what those reviewers receive and shifting the compliance conversation from the end of the content lifecycle to the beginning.
AI prechecks for the MLR review process
The core application is AI pre-screening, sometimes called AI prechecks for the MLR review process: automated checks that scan a draft for compliance issues before it enters the formal review queue. A pre-screening pass can flag off-label language, identify claims that lack a supporting reference, check that fair balance requirements are addressed, and compare draft copy against a centralised claims library. All of this happens before a medical, legal, or regulatory reviewer sees the asset. The practical effect is that reviewers spend their time on genuine judgment calls rather than catching preventable errors.
MLR review automation at this stage, before content reaches reviewers, is where the largest efficiency gains are available. The goal is not to automate the review decision itself, but to reduce the number of issues that enter review in the first place.
What the data shows
The potential of AI-enabled automation across regulated pharmaceutical processes is well documented. McKinsey has reported that leading pharmaceutical companies have compressed regulatory submission timelines by 50 to 65 percent through AI-enabled automation and workflow redesign, a finding that reflects the broader efficiency gains available when AI is applied systematically to compliance-heavy processes.3 While MLR promotional review differs in scope from drug submission dossiers, the underlying dynamic is the same: AI reduces the administrative burden on skilled reviewers, freeing their capacity for higher-order work.
The human reviewer remains essential
Regulatory accountability for promotional content rests with the company, not the technology. FDA guidance makes clear that human judgment and documented approval are required for regulated promotional materials. No AI system currently meets the standard required to substitute for that accountability. The most effective implementations keep humans firmly in the loop, using AI to eliminate administrative friction while preserving reviewer authority.
AI at the content creation stage
When compliance checks are integrated directly into the writing environment, flagging issues as content is drafted rather than after it is submitted, marketing teams produce cleaner first drafts. The compliance feedback loop, which traditionally ran on a cycle of days or weeks, can compress significantly. Teams get near-real-time compliance signals during the drafting process rather than waiting for a reviewer to flag problems two weeks later.
This is the model that tools like Caidera are built around. Rather than functioning as a post-production compliance scanner, Caidera integrates pre-screening directly into the content creation workflow, scanning draft content for potential issues against regulatory frameworks including HWG, AMG, MDR, and IVDR, and flagging concerns before they reach the MLR queue. For teams using this approach, the practical effect is typically fewer revision rounds and shorter overall cycle times, with the review process directed toward genuine judgment calls rather than correctable errors.
For a deeper look at how AI is changing the approval process, read our case study on how Caidera accelerates life sciences marketing.
AI will not make MLR review obsolete. But for teams managing growing content volumes under increasing regulatory scrutiny, it is rapidly becoming the difference between a process that scales and one that does not.
Frequently Asked Questions
What does MLR stand for?
MLR stands for Medical, Legal, and Regulatory. It refers to the cross-functional review process that pharmaceutical, biotech, and medical device companies use to evaluate promotional and marketing materials for scientific accuracy, legal soundness, and regulatory compliance before publication.
What does MLR stand for in medical terms?
In medical and pharmaceutical contexts, the MLR full form is Medical, Legal, and Regulatory review. The process ensures that all promotional content, from clinical claims to visual elements, complies with applicable regulations before it reaches healthcare professionals or patients.
What is a Promotional Review Committee?
A Promotional Review Committee (PRC) is the standing team responsible for conducting MLR review within an organisation. It typically includes representatives from medical affairs, legal, and regulatory, and meets on a regular cadence to process submissions. Some organisations use PRC and MLR interchangeably; others distinguish between the committee structure (PRC) and the review process itself (MLR).
How long does an MLR review take?
At mid- and large-sized pharmaceutical companies, review cycles commonly stretch to 50 to 60 days per content piece under standard workflows.1 Organisations that have optimised their processes through parallel review structures, standardised submissions, and digital workflow tools can reduce this significantly. The most common source of delay is not the review itself but incomplete submissions and fragmented feedback processes upstream.
What is the difference between MLR review and PRC review?
The terms are often used interchangeably. MLR describes the three disciplines involved: Medical, Legal, Regulatory. PRC describes the committee structure that conducts the review. In practice, an organisation might say their content goes through PRC review, meaning it has been assessed by their Medical, Legal, and Regulatory functions.
Can AI replace the MLR review?
No. AI can automate pre-screening checks, flag compliance issues before they reach reviewers, and reduce the number of revision rounds required, but it cannot substitute for the judgment, contextual interpretation, and accountability that human reviewers provide. FDA guidance requires documented human oversight for regulated promotional materials. The most effective AI implementations support reviewers rather than replace them.
Does MLR review apply to digital content?
Yes. MLR review applies to all promotional content regardless of format or channel, including social media posts, digital advertising, email campaigns, websites, and video content. The regulatory obligations do not change based on the medium. If content promotes a prescription drug, biologic, or regulated medical device, it requires review.
See How Caidera Automates MLR Pre-Checks. Reduce review cycles from weeks to hours, without replacing the human judgment your compliance process depends on. Book a demo | Explore the platform
Sources
1 Indegene — Pharma Content Compliance Report
2 Veeva — Content Benchmark Report (2023)
3 McKinsey — "Rewiring pharma's regulatory submissions with AI and zero-based design" (2025)
