Compliance & Regulation
5 articles

The Complete Guide to MLR Review
MLR review is the single biggest bottleneck in pharma marketing. This comprehensive guide covers what Medical, Legal, and Regulatory review means, how the process works, why it breaks down, and how AI is changing what is possible for life sciences content teams.

MLR Review: Why Your Team Waits Weeks — And How AI Changes That
The MLR process costs pharma teams up to 40 days per campaign. Learn how AI compliance tools like Caidera radically accelerate the review — without compromising quality.

Why Email Sabotages Your Marketing Approvals – And What Structured Workflows Do Better
Email-based approvals structurally fail in life sciences. Learn how structured workflows with AI pre-screening cut cycle times in half — without compliance compromises.
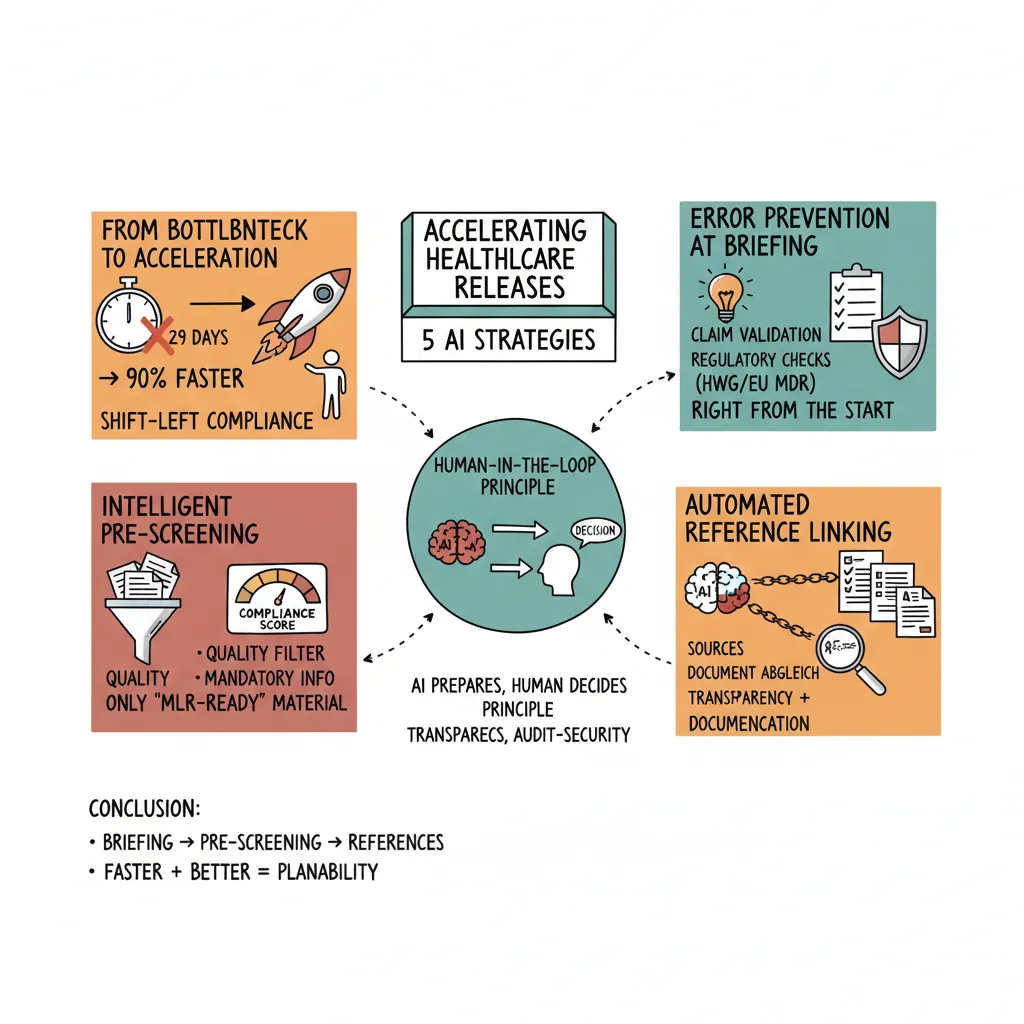
Accelerating Healthcare Approvals: 3 AI Strategies That Turn Weeks Into Hours
The MLR review takes an average of 29 days. Learn how AI-powered compliance checking reduces the approval process in pharma, medtech, and biotech by up to 90% while maintaining full regulatory compliance.

German Drug Advertising (HWG): Healthcare Compliance Guide
In the life sciences sector, marketing managers face the dual challenge of promoting innovative solutions while complying with strict regulatory requirements. The German Drug Advertising Act (HWG) is a fundamental set of rules governing advertising practices for pharmaceuticals and medical devices in Germany.